In the early 1960s, a series of case reports related zinc deficiency to stunted growth and sexual maturation in both male and female adolescents.
It turned out that zinc is kind of a big deal for growth and development.
Over 300 enzymes are zinc-dependent, reflecting the diverse biological functions which require this nutrient.
“Metalloproteins” are proteins in the body that bind to metal minerals [e.g., iron, copper, zinc], and up to 10% of the human genome encodes for zinc-containing proteins.
The issue with this important mineral is that we don’t know as much as we would like given the dizzying array of physiological functions that require zinc.
The Complexity of Zinc Homeostasis
The human body contains ~2-3g of zinc, and the processes regulating zinc homeostasis are complex.
Both zinc in cells [intracellular] and “free” zinc in the circulation [extracellular] are maintained in tight ranges, and zinc levels in the body are based on zinc requirements, i.e., greater absorption when lower levels are detected.
Zinc homeostasis is controlled by several factors: zinc transporters [ZTs], zinc importers [ZIPs], and metallothioneins [MTs], which are zinc-sensing molecules in the cell that regulate intracellular zinc levels.
Zinc is absorbed by ZIPs in the small intestine, and zinc levels are based on physiological needs and regulated at the level of absorption.
For example, when zinc levels in intestinal cells are high, concentrations of free, extracellular zinc are limited to maintain homeostasis.
Intracellular zinc is tightly regulated to prevent the adverse effects of either zinc deficiency or zinc excess. The expression of ZTs, ZIPs, and MTs, is regulated by zinc levels and is tissue-specific, i.e., different responses to zinc levels in different tissues.
When free zinc levels in the cell become elevated, MTs bind to that free zinc to maintain free zinc concentrations within tight ranges.
Intracellular zinc levels are also maintained by a process of transferring zinc within the cell [i.e., from the cytosol to organelles] or exporting zinc out of the cell.
All of these mechanisms are designed to respond to fluctuations in free zinc levels within the cell and maintain zinc homeostasis, which is the foundational starting point for understanding zinc and brain health.
Does Zinc Boost the Brain?
There is evidence that blood zinc levels may be lower in individuals with depression. In a 2013 meta-analysis by Swardfager et al., the severity of depression symptom scores increased as blood zinc levels decreased.
Consequently, it has been hypothesised that zinc supplementation may improve depressive symptoms, leading to specific research into zinc for depression. One proposed mechanism is that zinc supplementation may enhance levels of brain-derived neurotropic factor [BDNF], which is crucial for cognition.
A 2022 meta-analysis by Agh et al., including randomised controlled trials [RCTs] of zinc as a monotherapy, investigated the effects of zinc supplementation on BDNF levels.
They found, based on four RCTs, that zinc supplementation increased BDNF levels compared to placebo, with an effect size of 0.31 [95% CI 0.22 to 0.61]. Thus, the overall effect size for zinc supplementation was small, and the confidence intervals ranged from small to medium effect sizes.
Another recent meta-analysis of five RCTs of zinc supplementation and BDNF, which included all of the four studies included in the Agh et al. analysis, found a similar overall effect size of 0.31, but with a 95% CI range from -0.08 to 0.67, i.e., not statistically significant as the CI included 0.0.
Now consider that the effect size for exercise on BDNF in healthy individuals is 0.53 [95% CI 0.31 to 0.75], a medium effect size where the lower bound of the confidence intervals reflects the overall effect size of zinc supplementation.
Thus, if increasing BDNF levels is a desired outcome for cognitive function and health, there are more effective ways to achieve this end, in particular exercise.
What About Outcomes Over Mechanisms?
This is the question we always want to ask, so let’s consider the potential role of zinc on depression outcomes, independent of the effects of zinc supplementation on potential intermediate mechanisms, such as BDNF.
A 2012 systematic review by Lai et al., which included four trials, specifically examining zinc for depression, did not produce a quantified summary estimate of effect of zinc supplementation on depressive symptoms as the included studies were too heterogeneous to combine.
However, delving into some individual trials reveals a more interesting picture.
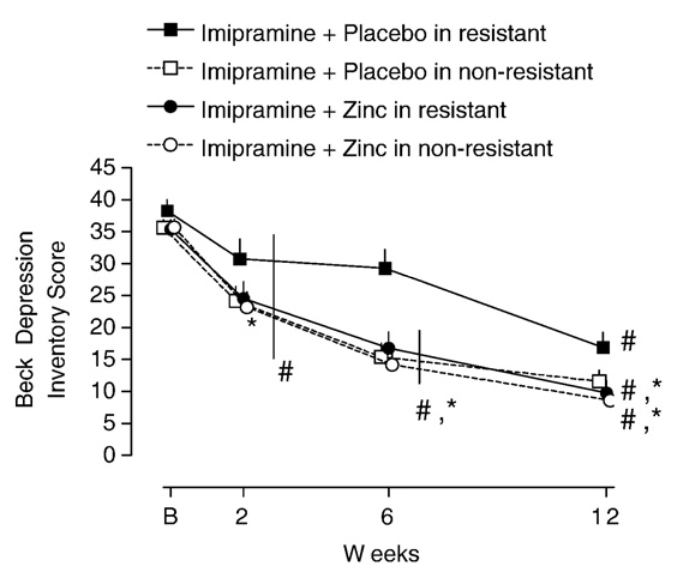
For example, a 2009 double-blind RCT from Siwek et al. compared the effects of zinc supplementation adjuvant to anti-depressant medication use in individuals both with and without anti-depressant resistance.
As you can see from the figure below, whether participants exhibited anti-depressant resistance or not, zinc supplementation led to significant reductions in depressive symptoms.
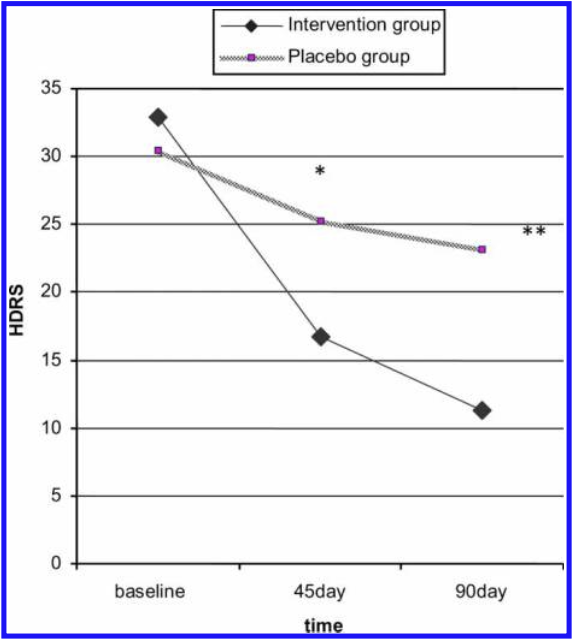
The most methodologically rigorous trial to date was published in 2014 by Ranjbar et al., which was conducted in participants all taking selective-serotonin uptake inhibitors [SSRIs] and with a diagnosis of Major Depressive Disorder [MDD].
Their trial demonstrated an independent effect of additional zinc supplementation adjuvant to SSRI in participants with MDD, as you can clearly see in the figure below.
This adjuvant effect of zinc supplementation was confirmed in a 2020 meta-analysis by da Silva et al., which found a small but significant effect size of 0.36 [95% CI, 0.04 to 0.67] in improving depressive symptoms in adults with MDD.
Final Thoughts
We must, however, conclude with a cautious tone, because there remain gaps in our understanding of zinc that warrant caution against unnecessary supplement use.
Plasma/serum zinc is primarily considered a biomarker for population-level zinc analyses, rather than individual-level, because at an individual level, changes in serum zinc status are not strong predictors of functional outcomes, e.g., growth.
Also, while zinc is important for the brain, it should be noted that two brain regions in which there are high zinc concentrations are also regions affected by plaque formation and neurodegeneration in Alzheimer’s Disease.
Both human and animal studies show high concentrations of zinc in amyloid plaque, suggesting zinc may promote beta-amyloid plaque development, which may be due to the overactivity of zinc transporters leading to higher concentrations of brain zinc.
The evidence to suggest zinc as an intervention for brain health is insufficient at this point.
There is, generally, little evidence to suggest a need for supplementation in otherwise healthy individuals, except for individuals who have an inadequate intake of dietary zinc.
In that context, proceed with caution: there appears to be little reason to supplement beyond 25-30mg/d zinc. Ultimately, more robust clinical trials are needed to fully map the boundaries of zinc and brain health.
Yours in Science,
Alan
Learn with Us.
You’ll find our most comprehensive resources in the Alinea Nutrition Education Hub.
Our weekly Deepdive takes a take a forensic look at a recent study: you’ll understand the background, the findings, and the relevance of the study in the context of the wider literature.
Our bi-monthly video Research Lectures condense complex topics into a visual presentation for you to maximise your learning experience.
And Exclusive Articles from researchers and academics in the field of nutrition science provide insights and perspectives from the people producing the research.
